
Especially important is the fact that the fundamental difference between simple asymmetric inductions and reactions taking place with spontaneous mirror symmetry breaking (SMBS) is now clear from a theoretical point of view, and this in spite of the fact that the specific reaction mechanisms of the few experimental examples on spontaneous mirror symmetry breaking (SMSB) are, until now, not well described. In the last few years, the role and mechanisms of natural chiral forces and chemical polarizations, able to lead to biases from the racemic composition, have been so well clarified that it is no longer a mystery that an important bias from the racemic composition can be obtained starting from achiral compounds. This, from a chemical point of view, is an unlikely scenario because it implies vast pools of enantiomerically pure mixtures of amino acids and sugars, and does not take into account the fact that racemization is likely to occur, at least to a certain degree, on the long timescales of evolution and for the experimental conditions necessary for the formation of the condensation of polymers. The origin of biological homochirality is thus considered as a separate and disjoint event: the formation of enantiomerically pure polymers is assumed to occur by starting from enantiomerically pure mixtures of their monomers. Models of mutualism and cooperative effects in autocatalytic sets, on quasi-species, the protein world and the RNA world (see for example, ) all assume tacitly the homochirality of the implied species. Homochirality is ubiquitous and ever present in biological chemistry from its very start, but there is a most curious absence of the chirality question in the studies concerning the abiotic stage of formation of the instructed polymers and replicators leading to the onset of Darwinian evolution, which is a striking paradox. However, such as has been quoted, in the study of the chemical evolution towards life processes, physical chemists have already paved the way for organic chemists, but have not succeeded in this task psychologically. Organic synthesis should be the experimental bench on which to test hypotheses on the emergence of life in abiotic terrestrial scenarios. In spite of the claims that chemistry should dedicate more efforts to study the origins of life and the fact that the number of research groups working on this topic is increasing, there is a lack of work compared to that generated in other chemical fields, the latter being arguably far less significant for the advancement of knowledge. Furthermore, when the emergence of biological chirality is assumed to occur during the stages of chemical evolution leading to the selection of polymeric species, one may hypothesize on a tandem track of the decrease of symmetry order towards biological homochirality, and the transition from the simple chemistry of astrophysical scenarios to the complexity of systems chemistry yielding Darwinian evolution. This could aid not only in the understanding of SMSB, but also the design of reliable scenarios in abiotic evolution where biological homochirality could have taken place. We review, in the first instance, which reaction networks lead to the nonlinear kinetics necessary for SMSB, and the thermodynamic features of the systems where this potentiality may be realized. (Shown on the orange graph below).Recent reports on both theoretical simulations and on the physical chemistry basis of spontaneous mirror symmetry breaking (SMSB), that is, asymmetric synthesis in the absence of any chiral polarizations other than those arising from the chiral recognition between enantiomers, strongly suggest that the same nonlinear dynamics acting during the crucial stages of abiotic chemical evolution leading to the formation and selection of instructed polymers and replicators, would have led to the homochirality of instructed polymers. This equation does not have y = x symmetry. 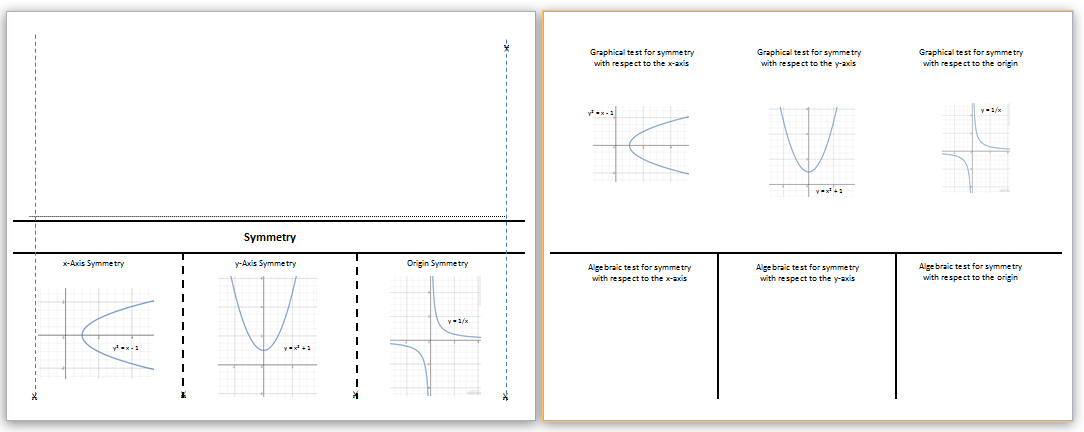 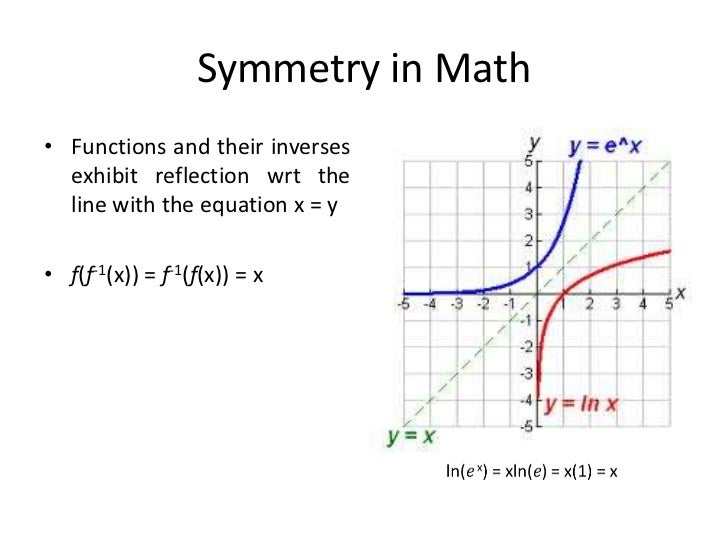
Symmetry y=x: (also known as diagonal symmetry) check for y = x symmetry by switching x and y (like we do to find inverses) and seeing if the equation remains the same. (Shown on red graph below.)īut, try typing y = 1/x into your graphing calculator. This equation has changed, so it does not have origin symmetry. Origin Symmetry: replace x and y with -x and -y respectively and see if the equation remains the same, if it does then the equation has origin symmetry. You can always check your answer by looking at the graph on a graphing calculator. X-axis Symmetry: replace y with -y and if the value of the equation does not change, then the equation has x-axis symmetry. We could easily alter this equation to have x-axis symmetry, though. Therefore, this equation does not have y-axis symmetry. If it does, then the equation has y-axis symmetry. Y-axis Symmetry: substitute -x for x in the equation and see if the equation remains the same (has the same value).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
